Cytology Solution
Pioneering advances in cervical disease prevention and cytology lab technology.

Setting a Standard
ThinPrep® is a worldwide standard for cervical sample collection. More than 1 billion ThinPrep vials have been used globally for cervical cancer screening.1 Just one patient sample is required for both cytology and molecular testing. We offer a comprehensive cytology solution, from sample collection to diagnosis, all within a complete suite of lab automation.
Improved Disease Detection
Our liquid-based cytology (LBC) test, the ThinPrep® Pap test, performs significantly better than conventional cytology in detecting diseases. Sensitivity and specificity are higher.2
Improved Visualisation
Moving to LBC results is a significant improvement in sample visualisation. The percentage of poor cell quality samples is decreased and sensitivity for the diagnosis of high-grade lesions is improved.2
Multiple Results From One Sample
Our cervical health solution enables clinicians to integrate cytological and virological (including HPV) results without the need to duplicate sampling and medical visits. This delivers greater efficiencies and the possibility of earlier detection.
Advancing the Early Detection of Cervical Cancer
250+
peer-reviewed studies confirming ThinPrep performance3
up to 300
slides prepared in 12h with ThinPrep® 5000 Processor Autoloader4
45 min
process for rapid cell block preparation5
A Comprehensive Cytology Solution
The ThinPrep Pap test is just one of several offerings in the ThinPrep system. Our ThinPrep processors, imagers and review scopes help improve laboratory workflow and help cytotechnologists to identify abnormalities. In addition, we are committed to providing laboratories with innovative and effective cytology solutions for non-gynecological testing needs. Explore the products in our solution here.
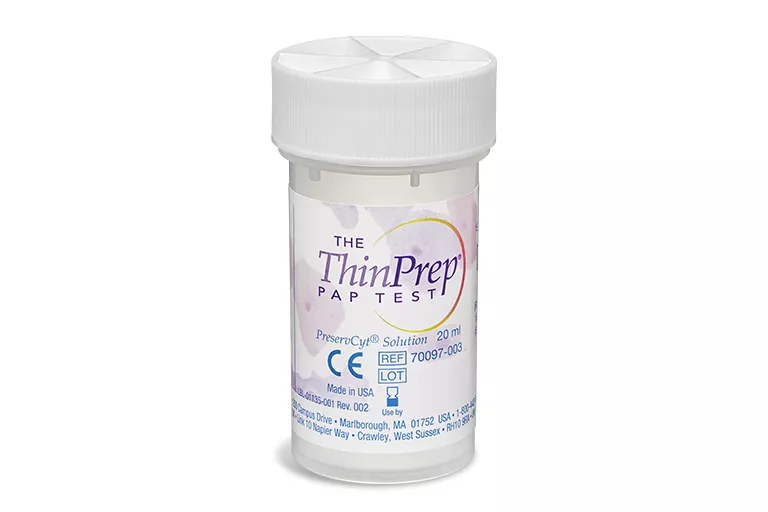
ThinPrep® PreserCyt Collection Vials
The ThinPrep® Pap Test is a worldwide standard for cervical sample collection and preservation trusted by healthcare professionals around the world. More than 1 billion ThinPrep vials have been used globally for cervical cancer screening. Only one single patient sample is required for both cytology and molecular testing.6
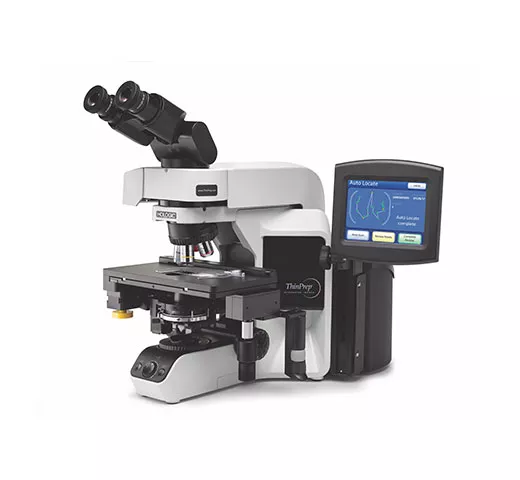
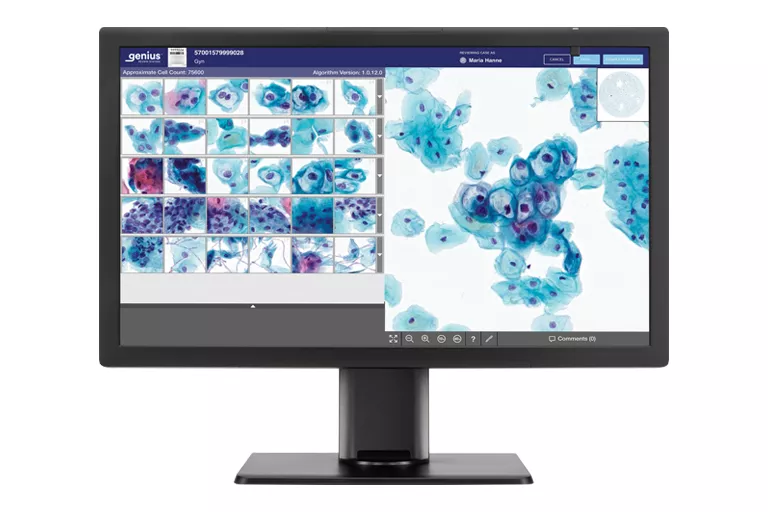
ThinPrep® Integrated Imager
Our integrated imager combines state-of-the-art imaging technology with the convenience of dual slide review in a single, automated, stand-alone microscope that fits neatly on a desktop. The dual review process combines human expertise with the power of computer imaging to deliver a superior solution for your lab.10

Cellient® Automated Cell Block System
This is the only fully automated cell block system and has an overall processing time of less than an hour. It uses vacuum-assisted filtration to rapidly concentrate available cells within the block. The system consistently produces diagnostically useful, high quality blocks for efficient review.5

Compass™ Stainer
With a smaller footprint, the Compass Stainer is perfect for low volume imaging labs using the Integrated Imager, or for labs that want a dedicated non-gyn stainer. It can store up to 15 staining protocols or programs. Protocol 1 is pre-loaded with the ThinPrep Stain Protocol for customer convenience.11
Insights
- Data based on Hologic sales numbers since launch in 2012 to 31Jan2020. File reference: AHPVtotalnumberJan2020 (Page 6 https://hologic.box.com/s/w5ccn2nza52n5010pr42ivy4ebfqbgxb) "smartCard-inline")
- Ikenberg et al, Screening for cervical cancer precursors with p16/Ki-67 dual-stained cytology: results of the PALMS study. J Natl Cancer Inst; 2013; 105: 1550–1557
- https://healthdxs.com/en View Published Data
- ThinPrep 5000 Processor AutoLoader, User Manual AW-22290-002 Rev 003
- Cellient Automated Cell Block System User Manual. MAN-08346-002 Rev 002
- ThinPrep® Pap Test PreservCyt Solution, Instructions for Use AW-22719-001 Rev 001
- Genius Digital Diagnostics_CE Mark Press Release_FINAL_11.15.20
- ThinPrep 5000 Processor User Manual, MAN-07493-002 Rev 003
- ThinPrep Genesis Processor, User Manual MAN-08098-002 Rev 002
- ThinPrep Integrated Imager, User Manual AW-22850-001 Rev 002
- Compass Stainer, User Manual AW-23958-001 Rev 001
Related Portfolios & Solutions

Molecular Scalable Solution
A scalable portfolio combining a broad, high performing assay menu with high throughput automation. Designed to flexibly scale to meet your needs, from a single patient result to population level screening.

Cervical Health
We pride ourselves on being champions of women’s health and global leaders in screening. From HPV to cytology, and now also AI based digital diagnostics, we offer a comprehensive and unique screening portfolio, from sample collection to diagnosis.
2797
Hologic BV, DA Vincilaan 5, 1930 Zaventem, Belgium
Notified Body number wherever applicable