STI Solution
Protect and improve reproductive and sexual health with early detection of sexually transmitted infections (STIs).
STI Testing for Improved Sexual Health
The Hologic STI Solution supports sexual health testing with a comprehensive, flexible menu of CE-marked STI assays. Our tests provide excellent sensitivity and specificity across a wide variety of targets. This instils confidence in diagnostic decisions and helps stop the spread of disease.
Test. Detect. Differentiate.
The Hologic STI solution combines a comprehensive assay menu with advanced automation. Consolidate your STI testing with fully automated, sample-to-result processing on the Panther® System.
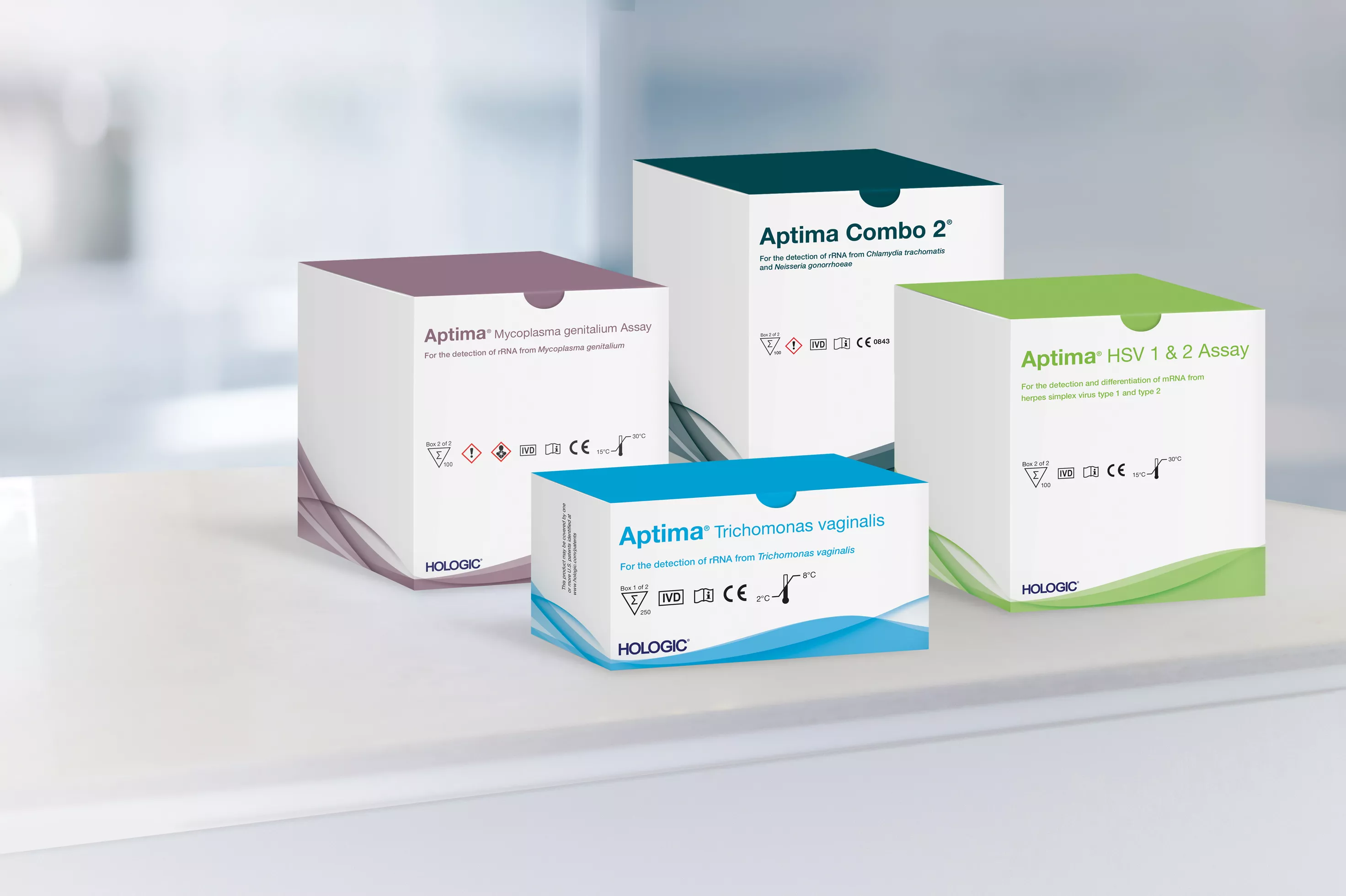
Aptima Combo 2® Assay (for CT/NG)
Protect and improve reproductive and sexual health through accurate and early detection of Chlamydia trachomatis (CT) and Neisseria gonorrhoea (NG). The dual target assay provides excellent diagnostic performance across a broad range of specimen types, from both patient and clinician collected samples.1

Aptima® Herpes Simplex Viruses 1 & 2 Assay
Expand your testing capability with qualitative detection and differentiation of Herpes simplex viruses (HSV) 1 and 2. A Nucleic Acid Amplification Test (NAAT) is the preferred testing method for genital herpes diagnosis.2,3 Distinguishing between HSV 1 and 2 is recommended for all patients with first-episode genital herpes.4

Aptima® Mycoplasma genitalium Assay
Protect sexual health through accurate and early detection of a prevalent and often overlooked STI, Mycoplasma genitalium. Emerging data has brought a new understanding of the prevalence and clinical impact of this bacterium, an infection that is often asymptomatic.5 NAATs that identify nucleic acids from Mycoplasma genitalium are the only useful methods for diagnosing infections.6, 7
Aptima® Trichomonas vaginalis Assay
Global prevalence of Trichomonas vaginalis (TV) infection is estimated to be higher than chlamydia and gonorrhoea combined.8 Clinical presentation is an unreliable indicator of TV infection in women, as 10-50% of infections are asymptomatic.9 The Aptima TV assay delivers up to 100% detection in both symptomatic and asymptomatic patients.10

Panther® Scalable Solution
The Hologic high-throughput solution allows you to expand your testing menu while adding on flexibility, capacity and walkaway time. The Panther® System is the foundation of Panther Scalable Solutions. It lets you customise your testing by choosing from a broad assay menu and instrument add-ons, allowing an economical and scalable path to your growth.
Aptima® Chlamydia trachomatis Assay
The single target assay provides excellent diagnostic performance across a broad range of specimen types, from both patient and clinician collected samples. This assay adds additional testing flexibility by using an alternative target region to the Aptima Combo 2 assay (for CT/NG), providing more confidence in the diagnosis.11,1
Aptima® Neisseria gonorrhoeae Assay
The single target assay provides excellent diagnostic performance across a broad range of specimen types, from both patient and clinician collected samples. This assay adds additional testing flexibility by using an alternative target region than the Aptima Combo 2® assay (for CT/NG), providing more confidence in the diagnosis.12,1
Insights
Simplify & Scale the Future of Diagnostics
The STI Solution is part of the Hologic Molecular Scalable Solution, a portfolio combining a broad, high performing assay menu with high throughput automation. Designed to flexibly scale to meet your needs, from a single patient result to population level screening.

Scale Your Operation
Explore how the Panther System helps you optimise today and prepare for tomorrow. The fully automated system loads samples in any order at any time, eliminates batch constraints and decreases turnaround time.
- Aptima Combo 2 Assay. [package insert]. AW-19693-001 Rev. 001. Hologic, Inc., 2020.
- Aptima Herpes Simplex Viruses 1 & 2 Assay [package insert]. AW-23071-001 Rev 001. San Diego, CA: Hologic, Inc., 2023.
- WHO Global Health Sector Strategy on Sexually Transmitted Infections 2016–2021. Accessed August 16, 2022. https://apps.who.int/iris/bitstream/handle/10665/246296/WHO-RHR-16.09-eng.pdf?sequence=1.
- Patel R, Kennedy O, Clarke E, et al. 2017 European guidelines for the management of genital herpes. Int J STD AIDS. 2017Dec;28(14):1366-1379
- Taylor-Robinson D, Jensen J. Mycoplasma genitalium: from Chrysalis to multicolored butterfly. Clin Microbiol Rev 2011;24:498-514
- Jensen JS, Cuisini M, Gomberg M, et al. 2021 European guideline on the management of Mycoplasma genitalium infections. J Eur Acad Dermatol Venereol. 2022 May;36(5):641-650
- Aptima Mycoplasma genitalium Assay [package insert] AW-22788-001 Rev 001 Hologic, Inc., 2023.
- Rowly J, Vander Hoon S, Korenromp E, et. al. Chlamydia, gonorrhoea, trichomoniasis and syphilis:global prevalence and incidence estimates 2016. Bull World Health Organ. Aug 2019; 97:548-562
- Sherrard J, Ison C, Moody j. et.al. United Kingdom National Guideline on the Management of Trichomonas vaginalis 2014. Int J STD AIDS 2014;25(8):541-49
- Aptima Trichomonas vaginalis Assay [package insert] AW-23069 Rev.001 San Diego, CA; Hologic, Inc., 2023.
- Aptima Chlamydia trachomatis Assay [Package Insert] 502184EN Rev. 010. Hologic Inc., 2022
- Aptima Neisseria gonorrhoeae Assay [Package Insert] AW-22785 Rev. 001. Hologic Inc., 2023
Related Portfolios & Solutions

Molecular Scalable Solution
A scalable portfolio combining a broad, high performing assay menu with high throughput automation. Designed to flexibly scale to meet your needs, from a single patient result to population level screening.

Cervical Health
We pride ourselves on being champions of women’s health and global leaders in screening. From HPV to cytology, and now also AI based digital diagnostics, we offer a comprehensive and unique screening portfolio, from sample collection to diagnosis.
1434
2797
Hologic BV, Da Vincilaan 5, 1930 Zaventem, Belgium.
Notified Body number wherever applicable
EC Representative Information wherever applicable