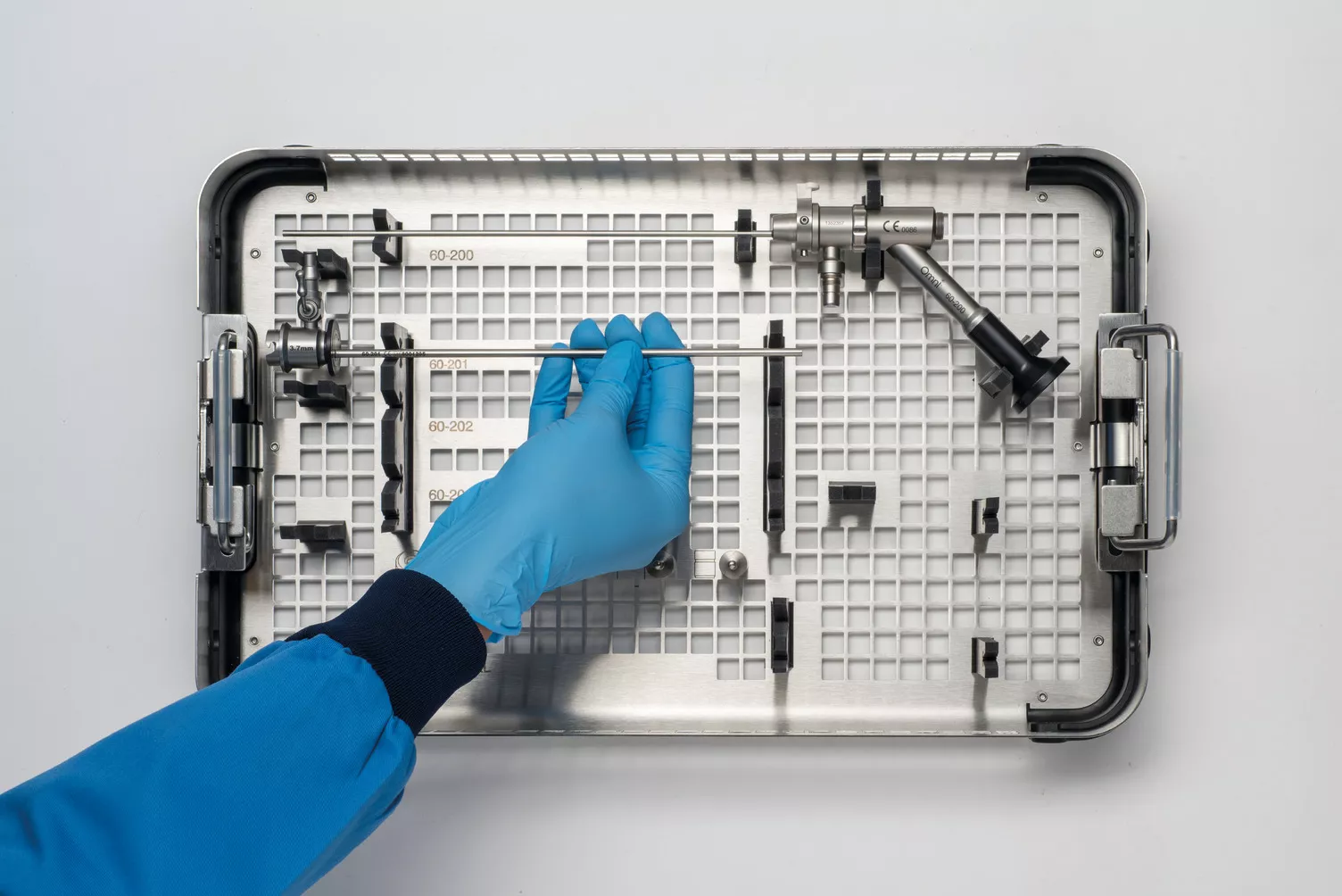
Omni® Hysteroscopes
High quality and flexibility help you maximise performance with an all-in-one scope.

Clear, Customised, Comprehensive
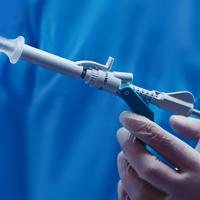
Omni Hysteroscopes offers three interchangeable sheaths with 0° or 30° options for flexible diagnostic and operative capabilities. A complete range that ensures you can adapt to each patient and the required procedure. Used in both an outpatient or operating room (OR) settings. Designed with you in mind to allow you to perform your best work.
See & Treat
Precise HD rod lens optics enables an optimal See & Treat experience.
Three hysteroscopes in one lets you see and treat pathology with a single hysteroscope.
Different Angles
Available with viewing angles of 0° and 30°. Together with the 3 interchangeable sheaths per angle and a longer working length of 200 mm.1
It enables optimised hysteroscopy with flexible diagnostic and surgical options.
Combine Tools
Optimising diagnostic and operative hysteroscopy with one versatile hysteroscope.
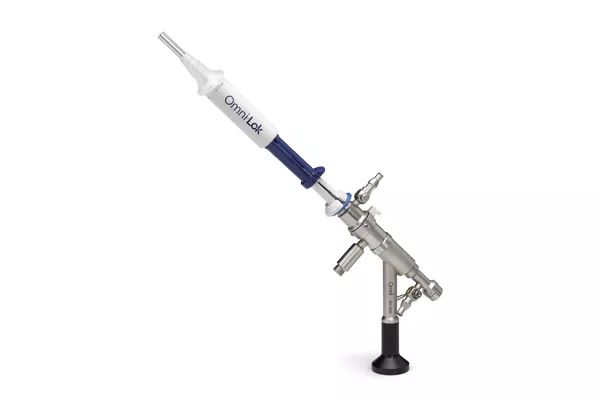
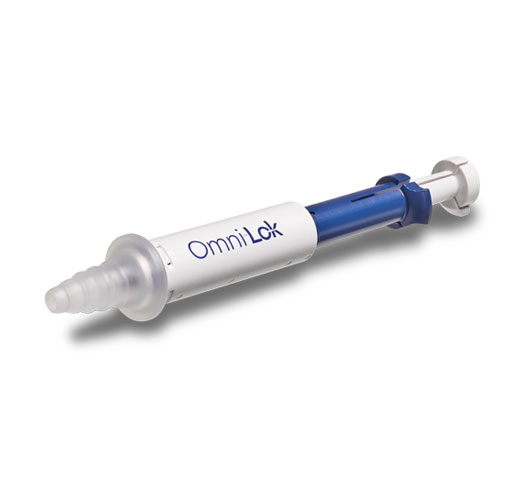
Omni scopes are compatible with a wide array of accessories, including Omni® Lok and Omni® 5Fr Seal which adapt to your preferred tools of choice.
Patient Comfort
Designed for greater patient comfort with easier insertion and small diameter sheaths.
Long, 200mm working length facilitates easier access and treatment for larger patients.
Improving Patient Care is Within Your Control
For many women, the diagnosis and treatment of uterine disorders is a long, painful and frustrating journey. Our team pioneers gynaecological diagnostic and surgical solutions that give professionals greater confidence in patient diagnosis and more choice over treatment options.
Omni Hysteroscopes are part of the Hologic Uterine Health Portfolio.

Confidence from Quality Rod Lens Visualisation1
1. Continuous flow with removable outflow channel.
2. Seal design helps prevent fluid splash.
3. Flexibility to support multiple light source attachments.
4. Operative sheath with 6.0 mm outer diameter.
5. Operative sheath with 5.5 mm outer diameter.
6.Diagnostic sheath with 3.7 mm outer diameter.
7. 0° and 30° angle, each with 80° field of view, provides a wide view of the uterine cavity.
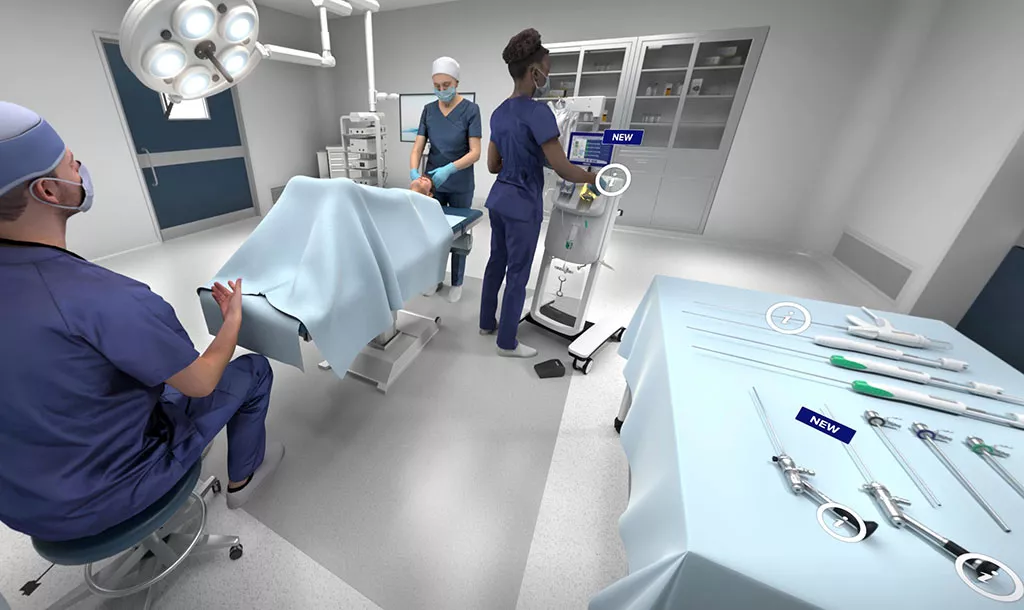
Visit Our Virtual Hospital
See how our Uterine Health products work, in an outpatient and operating room (OR) setting, including a Perinatal Suite.
Explore the Hologic Hysteroscopic Family
The Omni Tray
Empowering Women with Knowledge
Especially when it’s about things people tend to skirt around, like heavy periods or uterine issues. It’s time to stop waiting and wondering in silence. It’s time to take the next step toward overcoming heavy periods. We have the information that can help.
Evidence. Insight. Collaboration.
Our education portal improves patient care through excellence in education, communication of clinical and scientific evidence, and partnerships with the healthcare community.
Insights
1. Omni Hysteroscope Instructions for Use: MAN-05586-9940 Rev.011. San Diego, CA: Hologic, Inc.; 2022.
2. Omni Lok Cervical Seal Instructions for Use: AW-21052-002 Rev. 001. San Diego, CA: Hologic, Inc.; 2020.
3. As compared to procedures completed without the Omni Lok cervical seal.
4. Hologic Data on file, VAR-08420 Rev.001, Physician questionnaire after video introduction to device; N=15.
5. Hologic Data on file, DHM-07810 Rev.001, bench test utilizing extirpated uteri; N=3.
6. Hologic Data on file, VER-09159 Rev.002, bench test utilizing a uterine model; N=22.
7. Hologic Data on file, VER-10319 Rev.003, RTM-00573, MISC-07864-EUR-EN.
Documents
Safety Data Sheets
Package Inserts
Related Products
2797
Hologic BV, Da Vincilaan 5, 1930 Zaventem, Belgium.
Notified Body number wherever applicable
EC Representative Information wherever applicable